
TERA/VCP/p97 supplies energy to this protein complex to extract misfolded proteins from the ER lumen and transport them to the proteasome for degradation. TERA/VCP/p97 has a critical role in ER-associated protein degradation (ERAD) though interactions with Derlin-1, VCP-interacting membrane protein 9 and UFD1/NPL4 complex 10. TERA/VCP/p97 also interacts with clathrin 8, which coats the endosome membrane. 
TERA/VCP/p97 is essential for cistern reformation from mitotic Golgi fragments 7. For instance, the budding process of transition vesicles from smooth and rough ER is ATP-dependent and requires TERA/VCP/p97 (ref. TERA/VCP/p97 contains ATPase catalytic domains and regulatory N-terminal and C-terminal domains it forms a hexameric structure and couples ATP hydrolysis to cellular activities 5. The name TERA is derived from its function in the membrane transfer from the ER to the Golgi apparatus. TERA/VCP/p97 is a member of the ATPase associated with various activities AAA+ protein family involved in diverse cellular functions. 4), which causes spinocerebellar ataxia type 3. 3), and the mutant form of a polyglutamine disease protein, ataxin-3 (ref. The TERA/VCP/p97 protein also interacts with a normal length polyglutamine (polyQ) tract sequence derived from a transcription factor, Brn-2 (ref. Mutations in the human TERA/VCP/p97 gene cause frontotemporal dementia associated with inclusion body myopathy and early-onset Paget disease 1 or familial amyotrophic lateral sclerosis 2, and TERA/VCP/p97 is now considered a major causative gene for frontotemporal lobar degeneration (FTLD). Transitional endoplasmic reticulum (ER) ATPase (TERA), also known as valosin-containing protein (VCP) or p97, is implicated in various neurodegenerative disorders. Taken together, our results provide a novel common pathomechanism in multiple polyglutamine diseases that is mediated by DNA repair function of TERA/VCP/p97. Consistently, the recovery of lifespan in polyglutamine disease fly models by TERA/VCP/p97 corresponds well to the improvement of double-stranded break in neurons. Mutant polyglutamine proteins impair accumulation of TERA/VCP/p97 and interaction of related double-stranded break repair proteins, finally causing the increase of unrepaired double-stranded break. Among multiple functions of TERA/VCP/p97, we reveal that functional defect of TERA/VCP/p97 in DNA double-stranded break repair is critical for the pathology of neurons in which TERA/VCP/p97 is located dominantly in the nucleus in vivo. Although normal and mutant polyglutamine proteins interact with TERA/VCP/p97, only mutant proteins affect dynamism of TERA/VCP/p97. 
Here we show that transitional endoplasmic reticulum ATPase (TERA)/valosin-containing protein (VCP)/p97 directly binds to multiple polyglutamine disease proteins (huntingtin, ataxin-1, ataxin-7 and androgen receptor) via polyglutamine sequence. The receiving side of the Golgi apparatus is called the cis face.It is hypothesized that a common underlying mechanism links multiple neurodegenerative disorders. (credit: modification of work by Louisa Howard) Several vesicles can be seen near the Golgi apparatus. 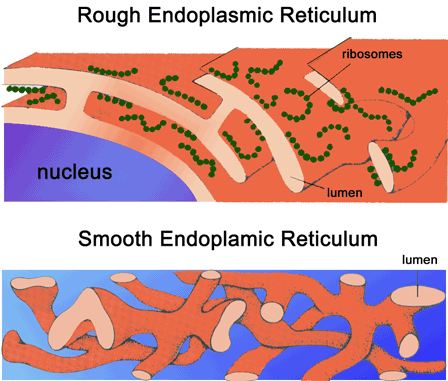
\): The Golgi apparatus in this white blood cell is visible as a stack of semicircular, flattened rings in the lower portion of the image.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
